Are we alone? Three words that make up a billion dollar question. Space agencies and organisations all over the world dedicate a lot of time, effort and money to attempting to answer this question. But what are they looking for exactly? What makes a place the ‘right spot’? In short, what are the requirements for life?
The issue is that we’ve only got one data point: life on Earth. We don’t want to assume that life elsewhere in this unimaginably large universe would necessarily resemble our own, but we also don’t know any aliens to compare notes with. So what we can do is analyse the requirements for life on Earth – energy (usually in the form of sunlight), carbon and liquid water – to see what makes them so important and then try to generalise them as much as possible.
Energy Source
Life on Earth’s surface uses the electromagnetic energy in sunlight as an energy source for photosynthesis (like plants), or eats things that perform photosynthesis (like rabbits) or eats things that eat things that perform photosynthesis (like us, minus vegetarians). Some life forms existing deep in the oceans, from ancient organisms living 3.8 billion years ago to tube worms today, are ‘chemosynthetic’ meaning that they use chemical energy as fuel rather than energy from the Sun.
In any case, life seems to need an energy source, although this source may differ. This can be seen as the most fundamental requirement from a physics perspective as life needs energy to maintain its own complexity and perform chemical reactions for its metabolism. Life is very ordered, complex and brimming with useful energy that can do work and is therefore what is known as a low entropy system. The universal, in general, is scattered and chaotic, its expansion spreading out its energy until it can’t do useful work and is therefore a high entropy system. The 2nd law of thermodynamics tells us that the entropy of the universe increases as time moves forward, meaning that things generally go from order to chaos. But this is not true of life, which emerged out of chaos and therefore goes against this general trend of increasing entropy. Just as you need energy when you swim against a strong current, life needs energy to go against this trend of increasing entropy and to maintain its own complexity*.
*Note: Life does not defy the 2nd law of thermodynamics, which states that the entropy of a closed system, such as the universe as a whole, must increase as time moves forward. The entropy on Earth is low, but to the universe the Earth is a miniscule speck in a small solar system in a modest galaxy. Entropy is allowed to decrease locally as long as it increases overall in the entire universe.
Carbon → Chemically Versatile Element for Building Genetic Structures
Carbon is a necessary element for all known life on Earth as it forms the backbone of the most important molecules. The carbon atom is light, containing just 6 protons, 6 neutrons and 6 electrons, meaning it is one of the first elements to be created in dying stars and consequentially is one of the most abundant elements in the universe. The great thing about carbon is its chemical versatility: it is the social butterfly of the periodic table, with each atom of carbon being able to bond with up to 4 other atoms. This enables carbon to link up with itself and many other elements to form the complex organic chains and structures vital to life including carbohydrates, lipids, proteins and nucleic acids such as DNA and RNA. Also, photosynthesis, the process that most life on Earth uses to harvest energy from the Sun, would not be possible without carbon dioxide, a compound generated by mixing carbon and oxygen.
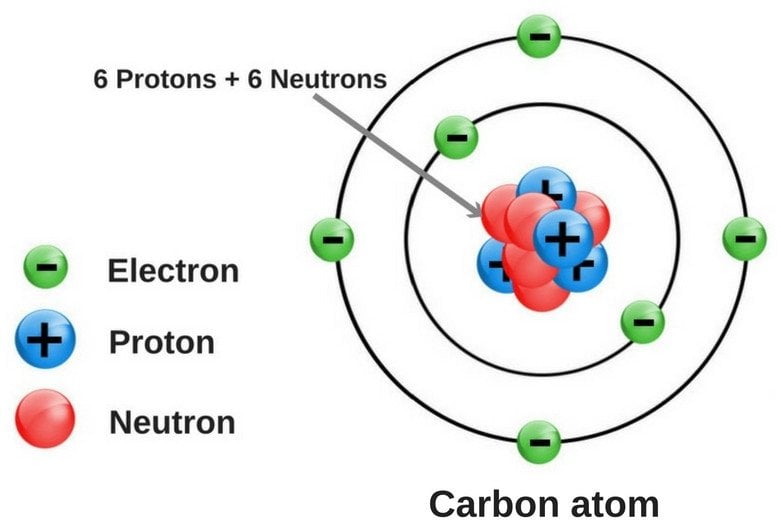
So why is carbon really so important and how can we generalise this? Carbon is vital to life on Earth because it is the backbone of the DNA structure, in which all information about an organism is laid out. All life, from single-celled bacterium to your pet dog to us, has one purpose in life: reproduction. Passing on your genetic material so that your species may survive is the fundamental, biological driver of living organisms and it can be imagined that although the process may differ across different life forms, this purpose is likely universal. Therefore, a chemically versatile element that can build life’s genetic material is possibly a general requirement for life. Silicon is a popular alternative to carbon as they are chemically similar.
Liquid Water → Polar Liquid
Liquid water is vital to all known life on Earth. We don’t know of any organisms that can thrive without water; some can enter a form of dehydrated hibernation called anhydrobiosis where their metabolism slows, but this is a temporary survival tactic and they only “come back to life” once they have access to liquid water. There are many reasons why water is so great for life on Earth:
1) Liquid over a wide range of temperatures – the presence of a liquid enables chemicals to dissolve and interact more readily with each other, increasing the efficiency and probability of the chemical reactions necessary for life. In a solid, particles aren’t able to move enough and in gases, they are too far apart and escape out of each other’s reach. A liquid is in just the right spot.
2) Good solvent – water can dissolve and pull apart many compounds, meaning life can easily snatch up the chemicals it needs from the spare parts.
3) Lipids don’t mix with water – cell membranes, which are made of lipids, are effective at retaining water within cells and making sure organisms don’t shrivel up.
4) High surface tension – this is why bubbles are round, why insects can walk on water and why you can balance a paper clip at the top of a glass of water, but more importantly it is the reason why water can travel up plants and blood can be pumped around our bodies against the force of gravity.
5) High heat capacity – heat transfer into and out of a body of water takes a long time; if there is a sudden change of temperature e.g. a heat wave or ice age, the ocean temperature will change slowly so marine life can be slowly acclimated.
6) Liquid water is denser than ice – this is unusual for a compound, but it means that glaciers float on top of oceans, which provided useful insulation for marine life living just underneath glaciers during ice ages. If water was normal and ice was denser than liquid water, the oceans would have frozen from the bottom up and there would have been no warm niches in which life could develop.
So what makes water have all these great properties for life? Almost all the properties that make water so suitable for life arise from the fact that it is a polar molecule: the two hydrogen atoms are slightly positive and the oxygen atom is slightly negative, meaning that there is an intra-molecular attraction within the water molecule and an inter-molecular attraction between water molecules. The inter-molecular attraction means that a group of water molecules are attracted together, therefore a lot of energy is required to break them apart – this is why water is a liquid over a broad range of temperatures and why it has a high heat capacity and high surface tension. The intra-molecular attraction means that the charged water molecule is good at pulling apart other polar particles, which is why water is a good solvent. From this analysis, it appears that a polar liquid could be another requirement for life. An alternative polar liquid to water could be ammonia.

Conclusion
Life on Earth needs: energy, carbon and liquid water. Energy is already a general requirement, but by studying what makes carbon and liquid water so useful to Earth life, we have been able to make a more general list of possible requirements for life, which are, in total: a source of energy, an element that can form the backbone of genetic structures and a polar liquid. At first, these may sound like quite a tall order, but in fact these ingredients could be satisfied within our very own Solar System, especially in the subsurface oceans of the icy moons of gas giants.
The header image is a photo taken 31 years ago by Voyager 1, on Valentine’s Day 1990, called “The Pale Blue Dot“. The “mote of dust suspended in a sunbeam”, as Carl Sagan called the Earth, is our home, caught from 6 billion kilometres away. That’s a healthy dose of perspective for today.




One thought on “Requirements for Extra-Terrestrial Life”